
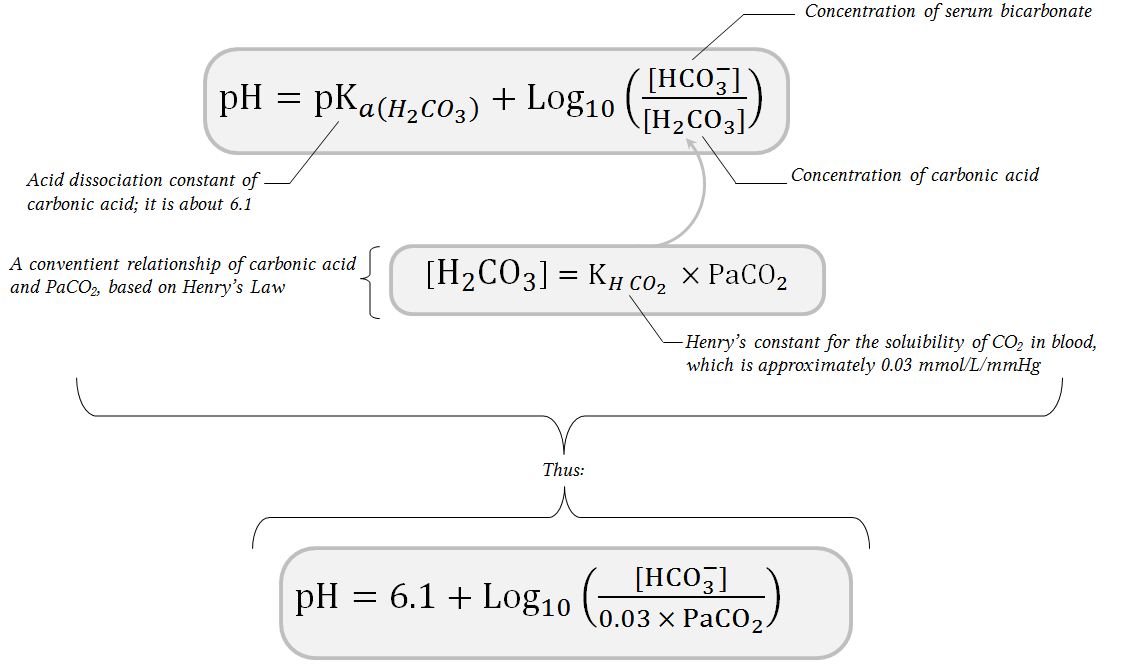
SOLVED: The pH of human blood is maintained at7.40 and is primarily controlled by the carbonic acid/bicarbonate (HCO/HCO)acid-base system.Use the HH equation to calculation the HCO3concentration if the HCO3 concentration is 24.5

SOLVED: Write an equation for HCO3- acting as an acid: HCO3- + H2O <-> H2CO3 + OH- CO3 + H2O <-> HCO3- + OH- HCO3- <-> H+ + CO3^2- HCO3- <-> H2CO3

Bicarbonate anion ( HCO3 ) - structural chemical formula and molecule model. Vector illustration Stock Vector Image & Art - Alamy

SOLVED: Calculate the concentration of H+, HCO3-, and CO3^2- in a 0.025 M H2CO3 solution. Note that H2CO3 is a diprotic acid. First ionization (Ka1 = 4.2 x 10^-7): H2CO3 (aq) ->

Can someone explain where did the CO3(2-) and HCO3(-) came from? And how do I write the equations with HCl and Na2CO3? : r/chemhelp
![SOLVED: The concentration of bicarbonate is calculated using the equation below. What is the concentration of bicarbonate for a pH of 7.0 and a PaCO2 of 40 mmHg? [HCO3] pH = 6.1 + SOLVED: The concentration of bicarbonate is calculated using the equation below. What is the concentration of bicarbonate for a pH of 7.0 and a PaCO2 of 40 mmHg? [HCO3] pH = 6.1 +](https://cdn.numerade.com/ask_images/6f18db30c9624fa99fdc4cda7c2f7592.jpg)
SOLVED: The concentration of bicarbonate is calculated using the equation below. What is the concentration of bicarbonate for a pH of 7.0 and a PaCO2 of 40 mmHg? [HCO3] pH = 6.1 +

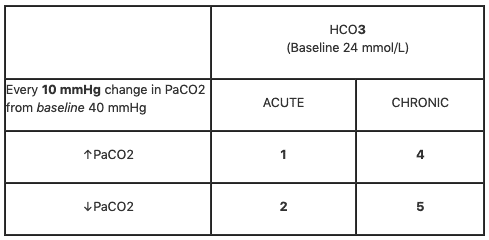
Rishi Kumar, MD - I created this table to teach my trainees how I approach acid-base problems assuming a normal bicarbonate (HCO3) of 24 mmol/L, PaCO2 40 mmHg, arterial pH 7.35-7.45, and
![PDF] Discrepancy between Measured Serum Total Carbon Dioxide Content and Bicarbonate Concentration Calculated from Arterial Blood Gases | Semantic Scholar PDF] Discrepancy between Measured Serum Total Carbon Dioxide Content and Bicarbonate Concentration Calculated from Arterial Blood Gases | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/d604591e54c0e843b180644b6e556990864a0b37/7-Figure4-1.png)

![Solved 3. calculate [H^+] based on the pH measurement in | Chegg.com Solved 3. calculate [H^+] based on the pH measurement in | Chegg.com](https://media.cheggcdn.com/study/973/9738f4a5-a8d9-4722-8a34-990ac1ca6db2/image.png)




![Solved Question 2 Calculate the blood [HCO3] of a patient | Chegg.com Solved Question 2 Calculate the blood [HCO3] of a patient | Chegg.com](https://media.cheggcdn.com/study/965/965de12f-e609-4ec1-80e3-f55348aade41/image)




![Solved Calculate the value of the ratios [HCO3-]/[CO32-] | Chegg.com Solved Calculate the value of the ratios [HCO3-]/[CO32-] | Chegg.com](https://media.cheggcdn.com/media/0b6/0b6129fb-dc7d-45f2-9788-1b56098b9225/phprZKjss)

