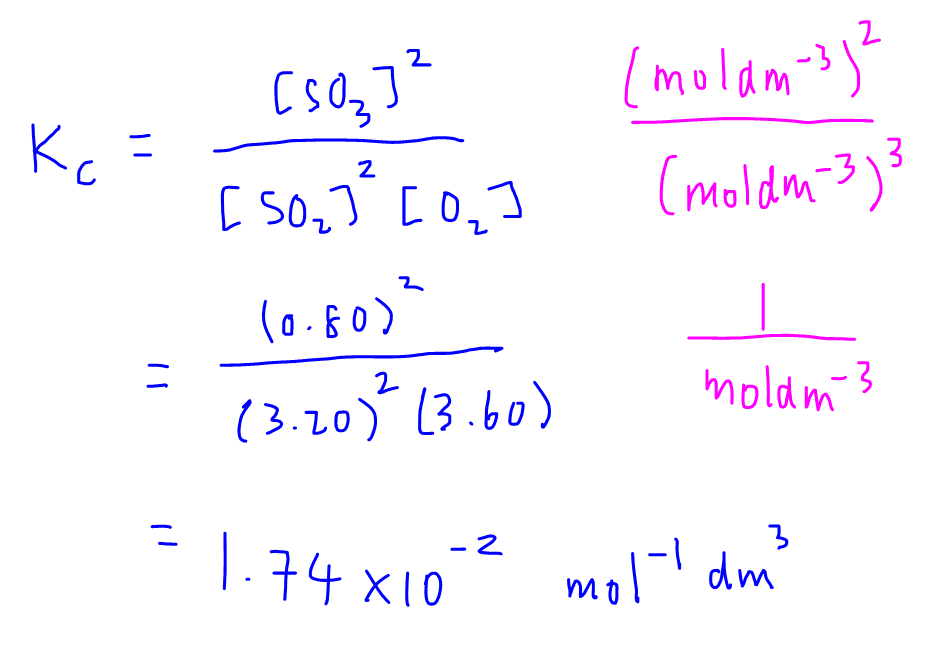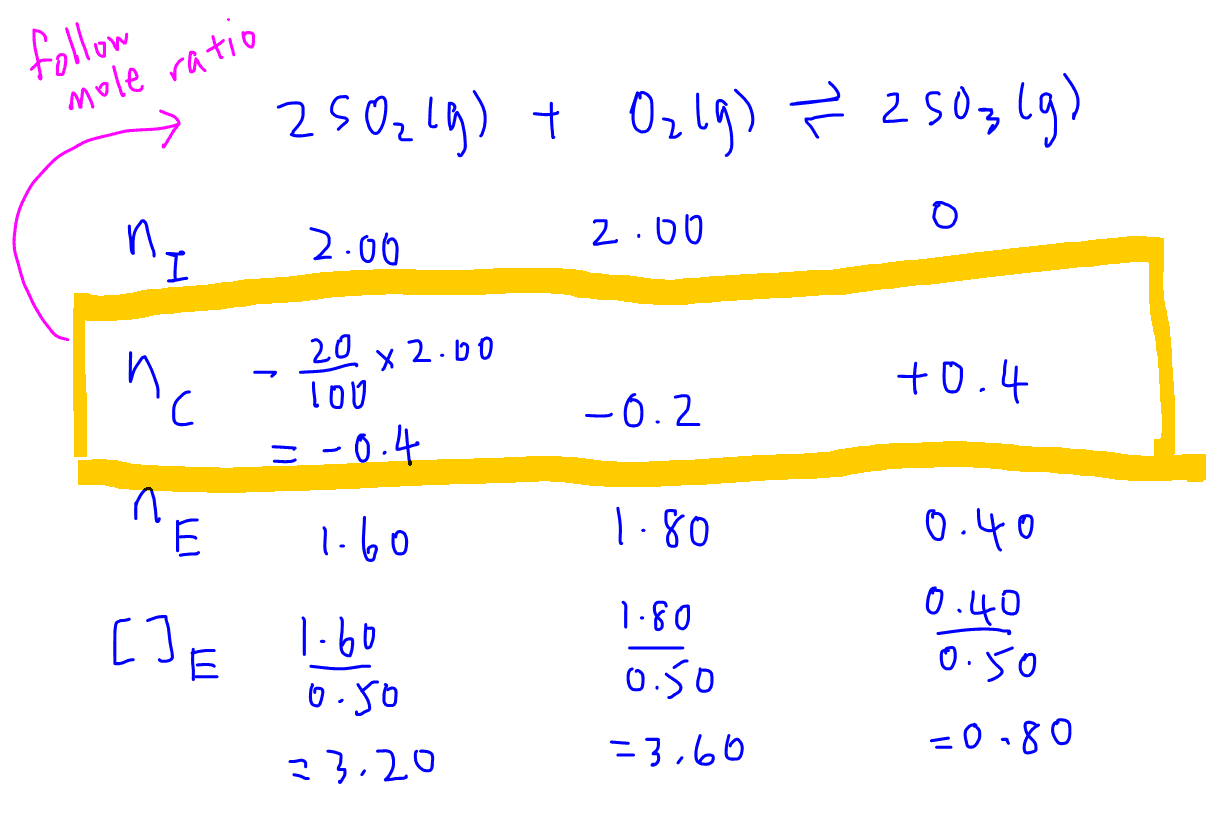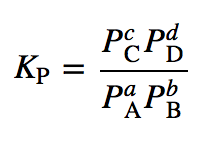A Simple Method To Calculate the Temperature Dependence of the Gibbs Energy and Chemical Equilibrium Constants | Journal of Chemical Education

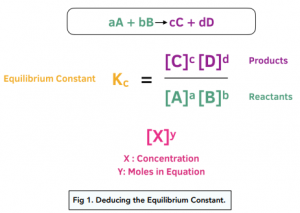
Learn how to calculate an equilibrium constant Kc. | Chemistry lessons, Teaching chemistry, Chemistry education
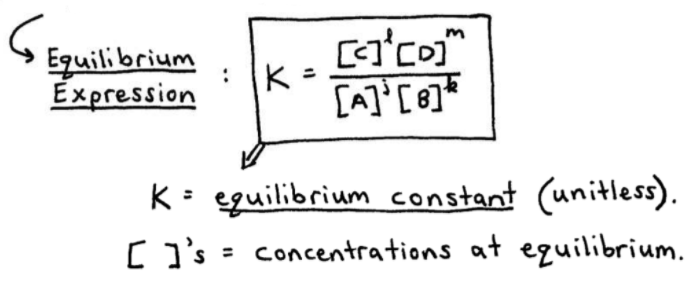
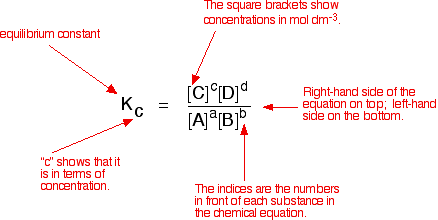
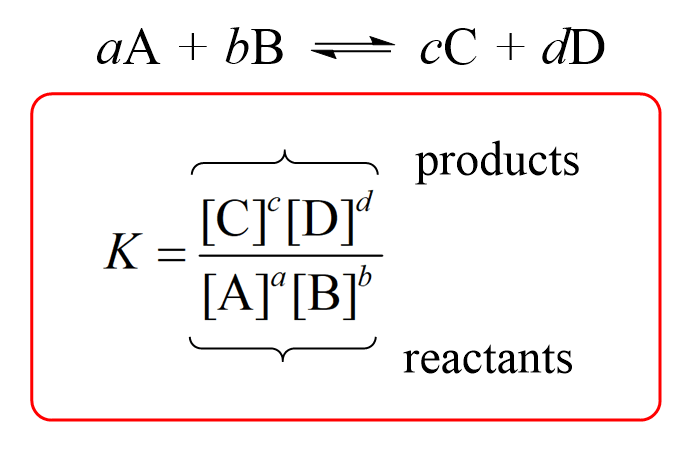
Equilibrium constant has a unit when the number of moles on both sides are not equal, why? If number of moles are not equal then there is no equilibrium then how equilibrium

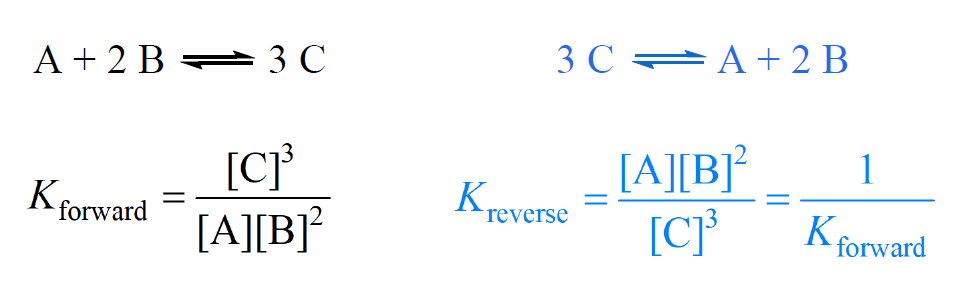
Easy tricks to calculate equilibrium constant based problems/Chemical eq... | Simple tricks, Equilibrium, Trick

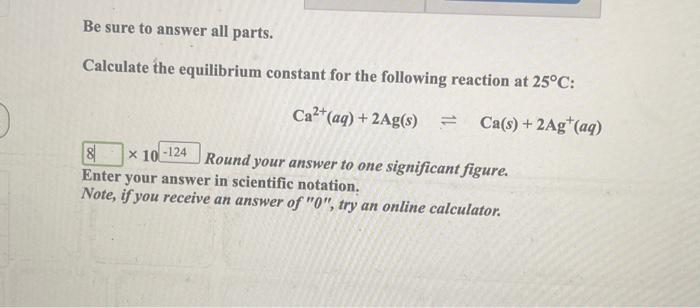
Calculating equilibrium constants from equilibrium concentrations or partial pressures (worked examples) (video) | Khan Academy