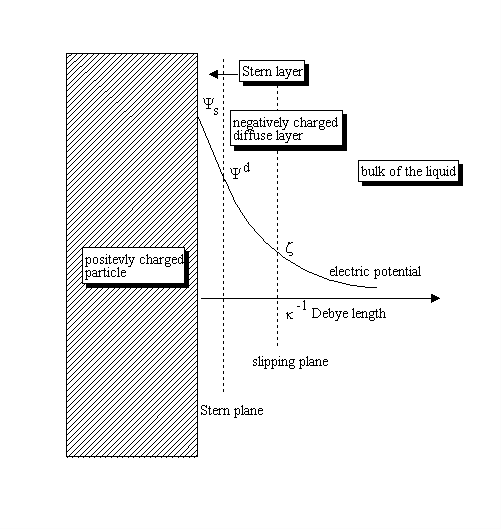
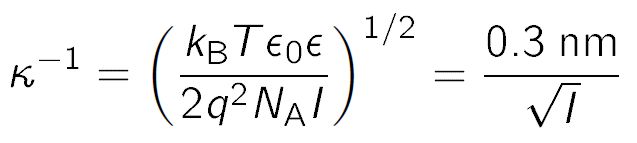SOLVED: a) Show that the Debye length can be approximated by (kT / (4Ï€e^2n0))^1/2, where T is the temperature in Kelvin and n0 is the number density in m^-3. b) Show that

SOLVED: HCl is a covalent molecule. The covalent bond in HCl has 24 % ionic character. The experimental dipole moment of this molecule is found to be 1.50 Debye. Calculate the bond

SOLVED: Compute the values of plasma pressure, Debye length, and the number of particles inside the Debye sphere for the following plasmas (n - cm-3, kT - eV). (90 marks) (a) Glow
![SOLVED: Equation List 1. Debye length - 1.304 nm for 1:1 electrolyte [NaCl] - 1.176 nm for 1.2 or 2:1 electrolyte [CaCl2] - 1.152 nm for 2:2 electrolyte [MgSO4] SOLVED: Equation List 1. Debye length - 1.304 nm for 1:1 electrolyte [NaCl] - 1.176 nm for 1.2 or 2:1 electrolyte [CaCl2] - 1.152 nm for 2:2 electrolyte [MgSO4]](https://cdn.numerade.com/ask_images/6fe865672fb949d8969b3329b257473c.jpg)
SOLVED: Equation List 1. Debye length - 1.304 nm for 1:1 electrolyte [NaCl] - 1.176 nm for 1.2 or 2:1 electrolyte [CaCl2] - 1.152 nm for 2:2 electrolyte [MgSO4]

Determination of Molecular Configuration by Debye Length Modulation | Journal of the American Chemical Society

Relation between the Debye length, ECV technique coordinate resolution,... | Download Scientific Diagram









![Solved where -1 ɛɛk]T к = Ze² Po Debye length if written as | Chegg.com Solved where -1 ɛɛk]T к = Ze² Po Debye length if written as | Chegg.com](https://media.cheggcdn.com/media/bbd/bbd52633-2636-4663-aa0f-a4b35e6852ec/php413gmk.png)